Incorporation of Zinc in Pre-alloyed CuIn[Zn]S2-ZnS Quantum Dots
Student: Jean Morales
Degree: M.S., July 2021
Major Professor: Dr. Colin Heyes
Research Area(s):
Nanoscience & Engineering
Background/Relevance
-
Commercially available CdSe quantum dots, QDs, are not suitable for bioimaging applications due to their high toxicity.
-
A solution to this issue is using CuInS2 /ZnS QDs which do not contain heavy metals and have longer fluorescence lifetime but controlling the blinking of these particles remains an issue.
Innovation
-
Incorporating zinc in pre-alloying steps to observe the behavior of the electronic and optical properties of CuInS2 QDs.
-
Establishing a direct correlation between the zinc composition of the QDs and blinking phenomena.
Approach
- Synthesis of the colloidal core QDs at 230ºC using 〖Cu〗^+, 〖In〗^(3+), 〖 Zn〗^(2+), and S^(2-) precursors, a high boiling point organic solvent and a ligand agent.
- Shelling process takes place at 230ºC and lasts from six to twelve hours.
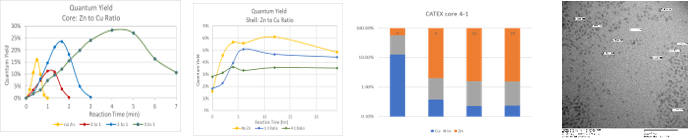
- Quantum yield is calculated from absorption and emission data.
- A fluorescence lifetime analysis is performed.
- Elemental analysis is done with ICP-MS.
- Characterization using a 100kV JEOL TEM.

Key Results
- Increasing the amount of zinc while synthesizing the core increases the quantum yield, QY, from 15% to about to 30%.
- Higher QY core QDs exhibited a smaller increase in QY upon shelling/cation exchange than those with no or little zinc.
- After 24 hr of cation exchange/shelling the QDs are composed of >98% ZnS.
- TEM size analysis shows heterogenous size distributions that increase as a function of shelling reaction time.
Conclusions
-
Incorporating zinc in the core synthesis step influences the electronic and optical properties of both alloyed core and post cation exchange.
-
The ratios of Cu and In change as the cation exchange reaction progresses.
-
Cation exchange of the core QDs yields a heterogeneous size distributions.
Future Work
-
Characterization of QDs using XRD and XPS.
-
Investigation of blinking properties.
-
Potential applications on time gated imaging, FLIM, bio tracking.
